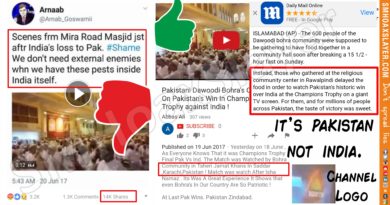
Did the US FDA sanction the widespread use of Remdesivir for treating COVID-19?
Multiple Facebook and Twitter posts claim that the United States Food and Drug Administration (FDA) has sanctioned the antiviral medicine named Remdesivir for treating COVID-19. The post was initially posted on Facebook and has since then been deleted. A screenshot of the post is follows and the archived post can be viewed here.


Archive.

 C
C
#remdesivir
FINALLY!
Good News:
Unang Gamot Laban sa Covid Aprubado ng US FDA
Remdesivir: Gusto Bilhin ng Maraming Bansa……— Rachelle S. Adarlo (@AdarloRachelle) May 9, 2020
The tweet translates into : “FINALLY! Good News: First Anti-Covid Drug Approved by the US FDA Remdesivir: Many Nations Want to Buy ……”
Truth
The above claims are misleading. The video posted initially that stated the US FDA has approved the medicine was taken from a CNN news segment. The video can be found here. Screenshots of the same are as follows:


Clinical trials have found that the drug shortens the recovery period of COVID-19 patients which resulted in FDA opening up the option for broader number of patients. However, though the FDA has allowed the emergency use of the drug, a statement issued on May 1 by Gilead Sciences says, “”Remdesivir has not undergone the same type of review as an FDA-approved or cleared product. There is no U.S. Food and Drug Administration (FDA) approved product available to treat COVID-19”. The entire press release can be viewed here and screenshots are as follows:


The FDA further confirmed that the drug is still an investigational one and is being only administered to certain emergency COVID-19 patients in a fact sheet that be viewed here. A screenshot is as follows:

- Is the video titled, “Amit Shah disowns Modi” being circulated on WhatsApp, a mobile-formatting virus? - June 7, 2020
- Was the White House stormed in by protestors amidst the death of African-American George Floyd? - June 4, 2020
- Video of a man made to lick spit in Surat shared with a communal angle - June 1, 2020